Introduction:
In the realm of colloidal science, accurately determining the zeta potential of particles in various environments is essential. However, when these environments feature high conductivity, standard measurement techniques face significant challenges. This article delves into these challenges and explores the advanced methodologies developed to overcome them, focusing on sterically-stabilized particles in high ionic strength solutions.
The Impact of High Conductivity on Zeta Potential Measurement:
-
Challenges with pH Measurement Accuracy:
- High Ionic Strength Effects: High salt concentrations/high conductivity in aqueous media can significantly affect the accuracy of pH measurements. This is primarily due to the reduced activity of hydrogen ions and alterations in the junction potential of Ag/AgCl reference electrodes.
- Implications for Zeta Potential: Since zeta potential measurements are closely tied to pH levels, inaccuracies in pH readings directly impact the reliability of measurements in high salt environments.
-
Limitations of Standard Electrokinetic Models:
- Steric Stabilizer Chain Interference: The presence of ionic groups within steric stabilizer chains at the particle-liquid interface challenges the validity of traditional electrokinetic models used for calculating zeta potential.
Methodological Advances to Address High Conductivity Challenges:
- Potentiometric Titration for Accurate pH Readings: Employing potentiometric acid-base titration techniques helps in adjusting pH measurements in high salt solutions, though issues arise in the presence of buffering actions by electrolytes.
- Analyzing Electrophoretic Mobility Across Electrolyte Concentrations: By studying the electrophoretic mobility of particles at constant pH over various electrolyte concentrations, more suitable electrokinetic models can be identified.
Study Insights:
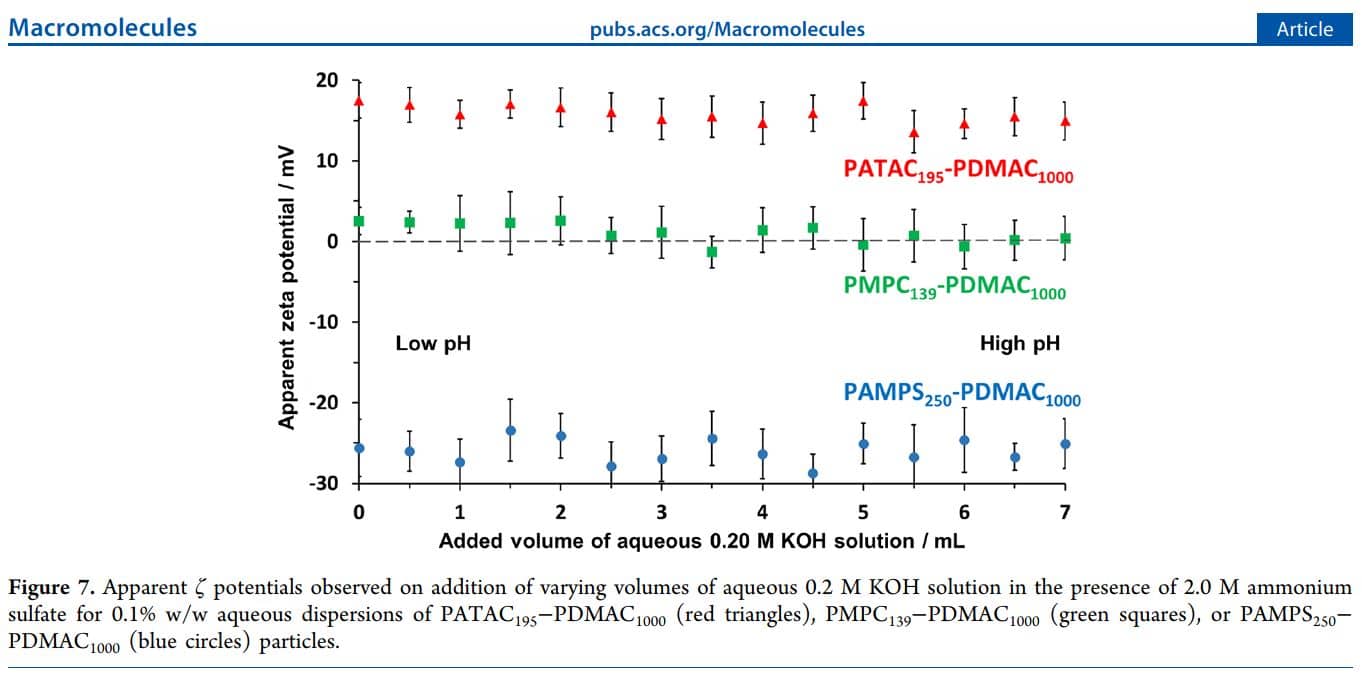
- Apparent vs. Theoretical Zeta Potentials: The study highlights discrepancies between apparent and theoretical zeta potentials in high salt conditions, particularly in solutions like 2.0 M ammonium sulfate. This contrast underlines the significant impact of ionic strength on zeta potential measurements.
- Particle Behavior Variability: Different particle types—cationic, anionic, and zwitterionic—demonstrate distinct zeta potentials in high conductivity solutions, influenced by the chemical nature of their steric stabilizer chains.
- Beyond Traditional Electrokinetics: Conventional electrokinetic theories based on non-conducting hard spheres fall short in explaining the behavior of these particles. Advanced models, such as the Hermans-Fujita model, offer a more accurate representation by considering factors like liquid flow through permeable spheres.
Conclusion:
This exploration into zeta potential analysis in high salt/high conductivity conditions reveals the complexities and intricacies involved in accurate measurement and interpretation. The findings emphasize the need for innovative approaches and models to tackle the unique challenges posed by high ionic strength and the properties of sterically-stabilized particles. As the field evolves, these insights will be crucial in advancing our understanding and application of colloidal science in complex environments.
You should see the original PDF in a viewer below but on Microsoft Edge you may see a Microsoft Defender SmartScreen warning. This is a false positive that has been reported to Microsoft. Please use the link above to access the PDF.